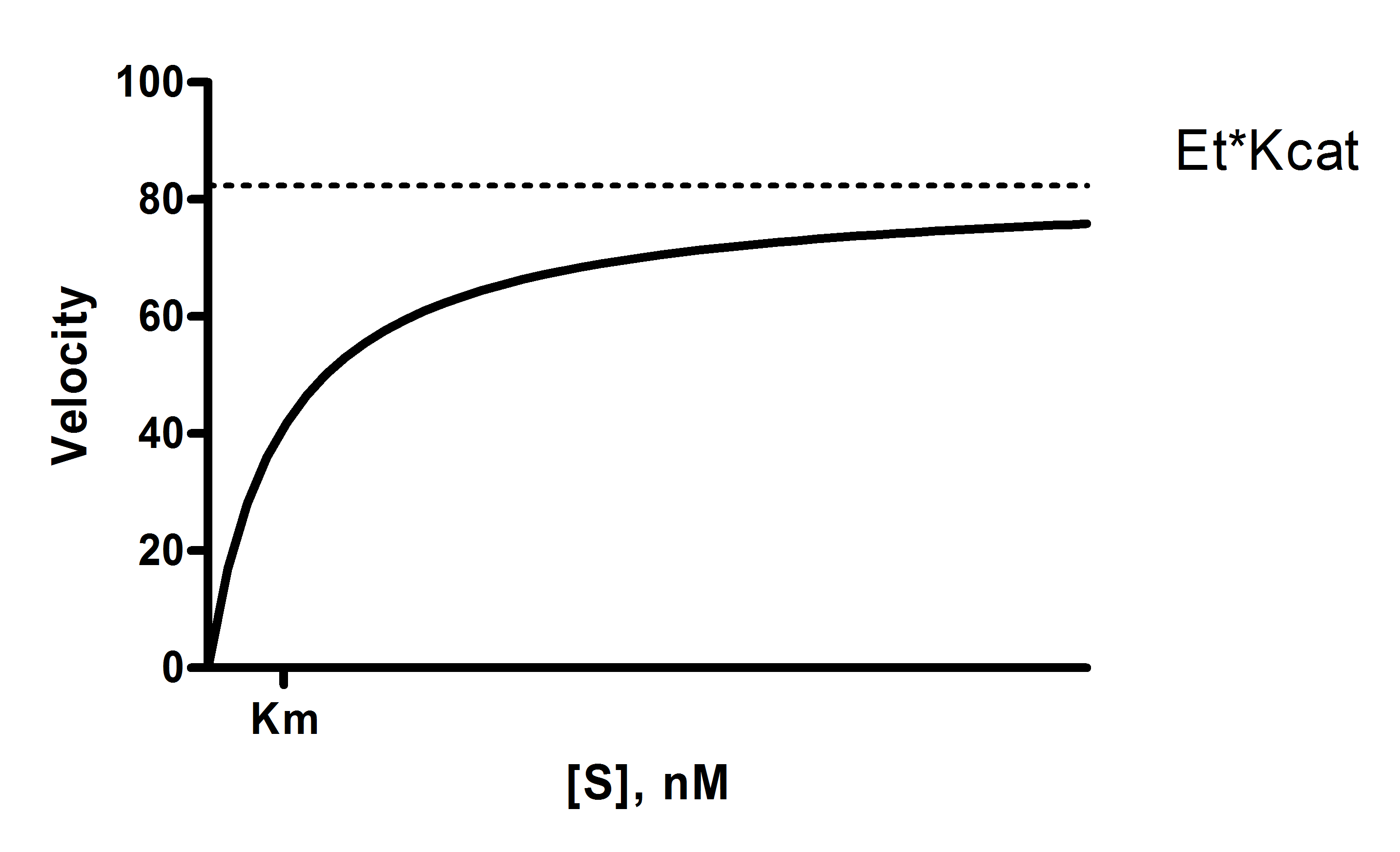
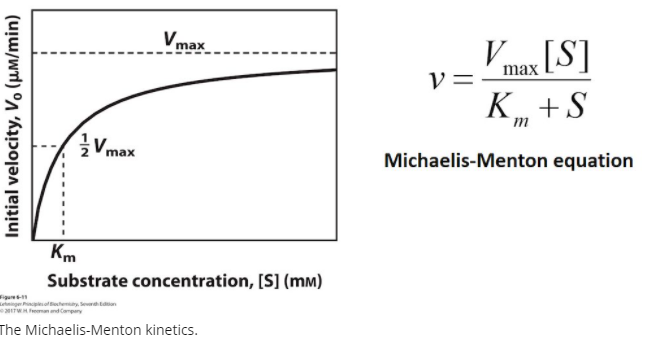
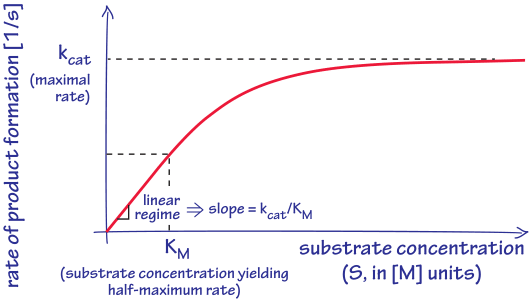
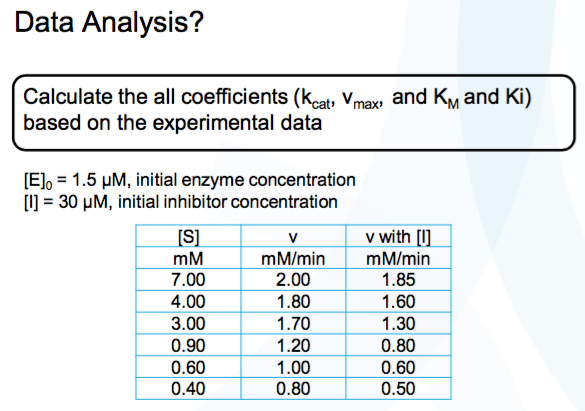
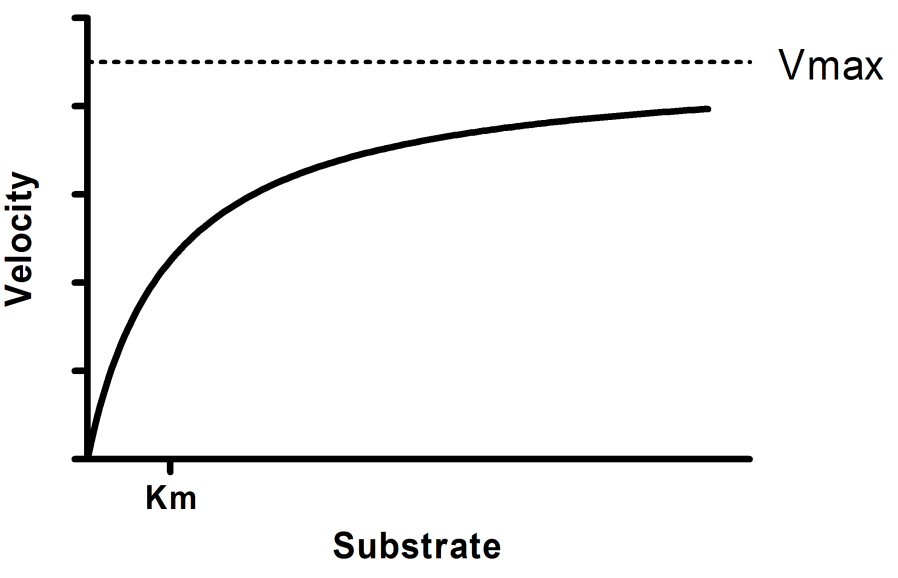
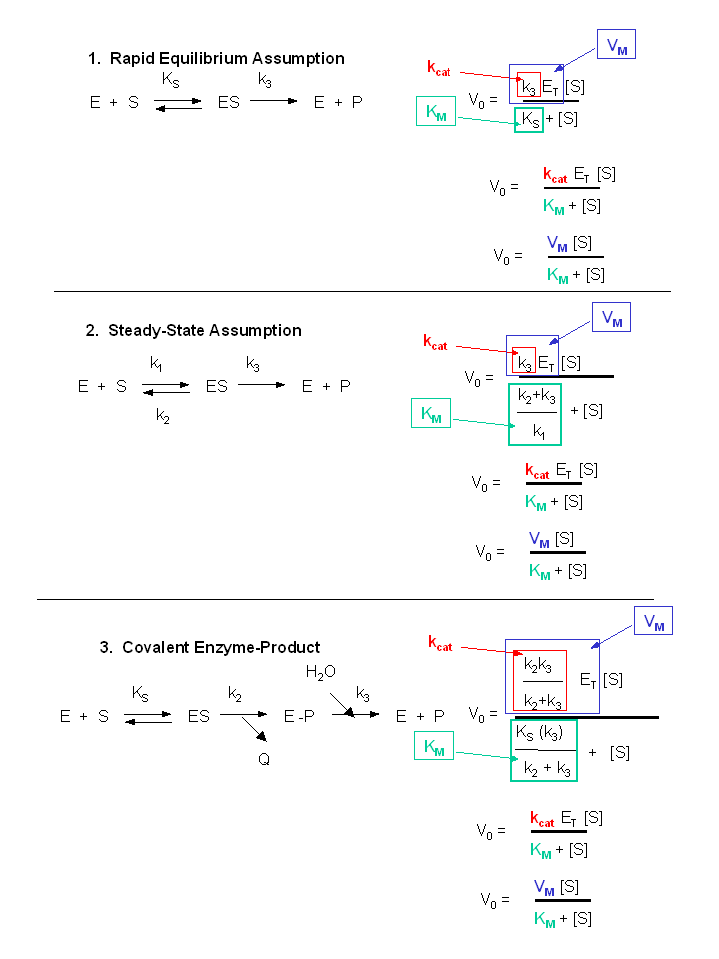
Structural Biochemistry/Enzyme/Michaelis and Menten Equation - Wikibooks, open books for an open world

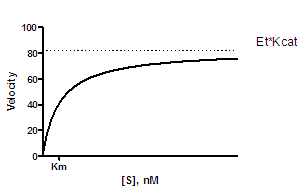
Michaelis–Menten plots for the calculation of the Km and Vmax values of... | Download Scientific Diagram

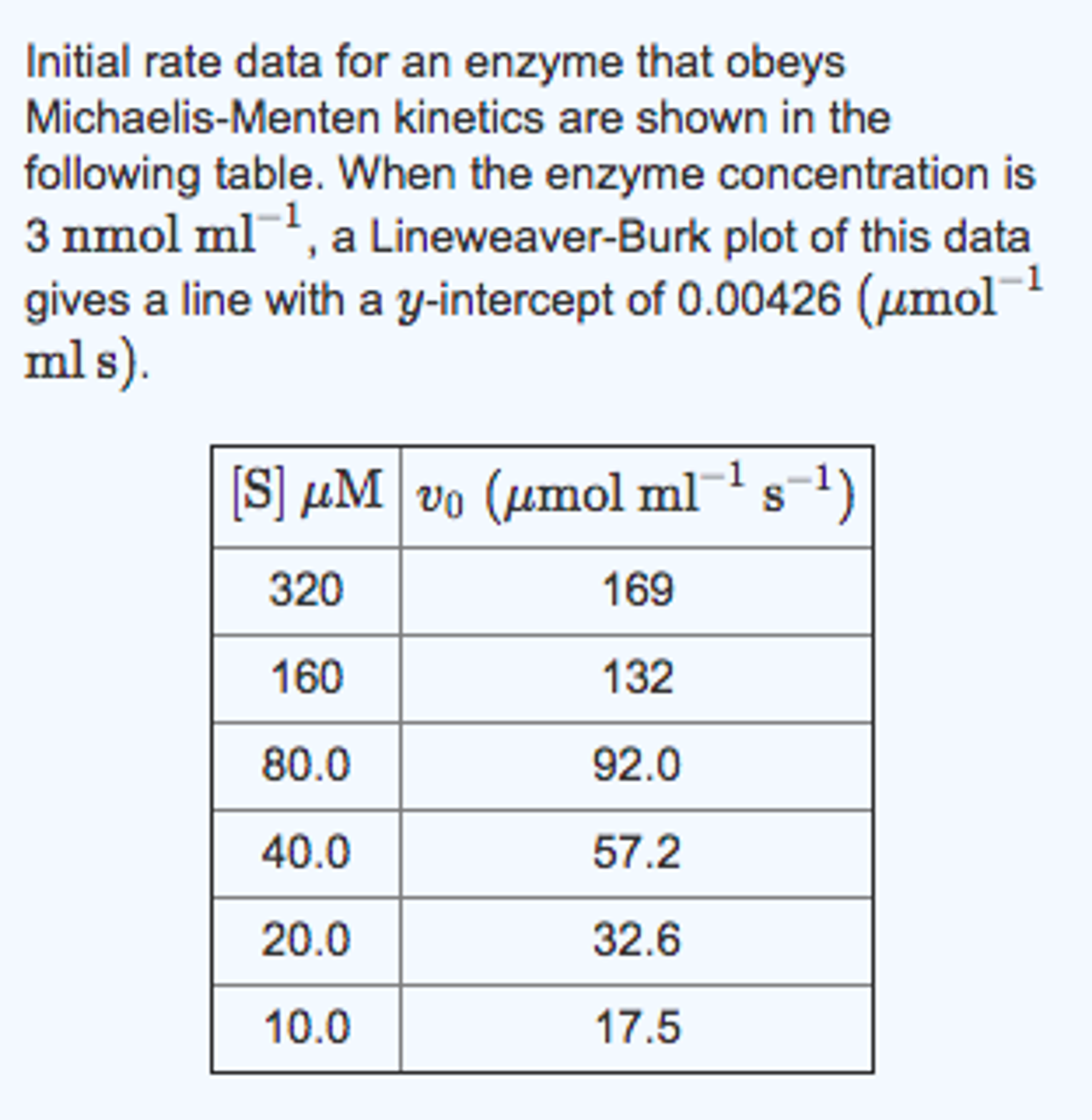
SOLVED: Using the Lineweaver-Burk plot calculate Vmax, Km and Kcat, 0.102 4= vtak 4210 Kcct 4. 79 " fhio Vrer 3.1ry Jrfan -9.1656 umfhv) Km - 0.486 Y 9.7656 Using the Michaelis-Menten

How to calculate kcat from Lineweaver-Burk plot following RFU? - Biochemistry and Molecular Biology - Science Forums
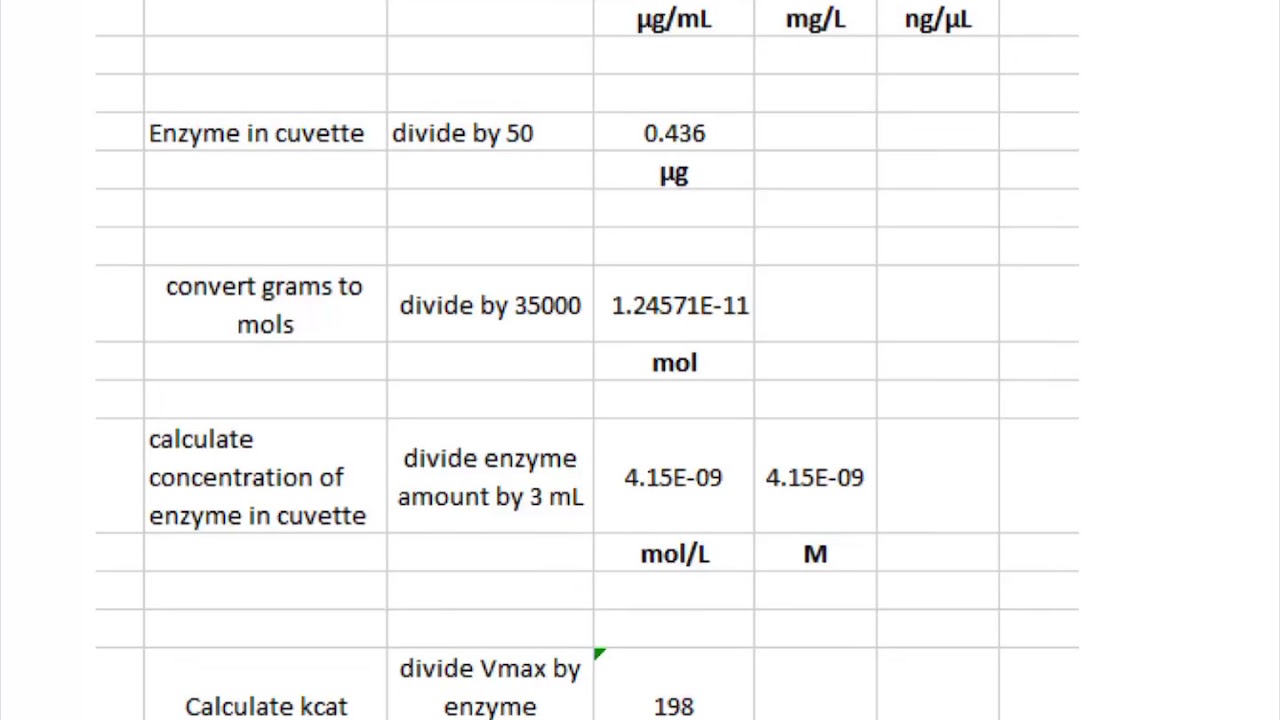
![SOLVED: Using your Vmax =2.225 umol/min from the uninhibited data, calculate kcat (kcat = Vmax/[ET], where ET is the total enzyme concentration. The concentration of stock solution of enzyme used was 0.35mg/mL SOLVED: Using your Vmax =2.225 umol/min from the uninhibited data, calculate kcat (kcat = Vmax/[ET], where ET is the total enzyme concentration. The concentration of stock solution of enzyme used was 0.35mg/mL](https://cdn.numerade.com/ask_previews/37951912-45bb-4020-9a65-3c82938b63de_large.jpg)
![Solved Show the derivation of Equation [9]. You may use the | Chegg.com Solved Show the derivation of Equation [9]. You may use the | Chegg.com](https://media.cheggcdn.com/media/f75/f75c5537-10cd-4ca8-a248-04c5112ae043/phpINRxM1.png)